The More Muscle, The Less Diabetes
If you have more lean muscle tissue, you have less risk of Type 2 Diabetes Mellitus (T2DM). Findings outlined by US and Chinese scientists provide further insights into how muscles use glucose in a feedback loop as well as its role in whole-body glucose balance.
“We found that skeletal muscle cells have machinery to directly sense glucose — in a certain sense it’s like the muscles can taste sugar, too,” said senior study author Dr Jiandie Lin, a faculty member at University of Michigan Life Sciences Institute commenting on his study published in Molecular Cell.
“The majority of cases of type 2 diabetes could be prevented by the adoption of a healthier lifestyle,” reported researchers led by Dr. Frank Hu of the Harvard School of Public Health in Boston, Massachusetts.
Widget not in any sidebars
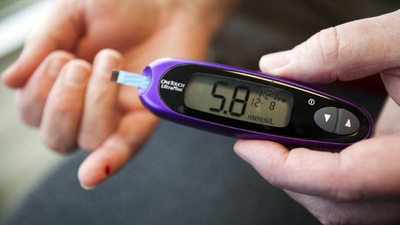
Raised blood glucose levels post-meal is the main symptom of T2DM. Elevated high blood sugar levels long-term (hyperglycaemia) can lead to serious health issues such as nerve damage or kidney failure.
One study looked at 13,600 people and followed them for 6 years. Researchers found that people who had just 10% greater amount of muscle tissue in their body, according to a special bioelectrical impedance measurement, had 12% less chance of developing pre-diabetes or diabetes type II.
The Diabetes Prevention Program (DPP), a trial published in 2002, found intensive lifestyle interventions such as diet or exercise were more effective than the diabetes drug metformin in preventing the onset of type 2 diabetes.
Reporting in a 2011 September issue of the Journal of Clinical Endocrinology and Metabolism, researchers from the David Geffen School of Medicine at UCLA suggest there is a correlation between greater muscle mass, relative to body size, and a substantially decreased risk of developing the metabolic changes that lead to diabetes.
Knockout Mice
Contributions of the glucose-sensing pathway in skeletal muscle by silencing an important gene–BAF60C–in cell cultures and in laboratory mice.
The team from the University of Michigan and Zhejiang University in China began looking at the contributions of the glucose-sensing pathway in skeletal muscle by silencing an important gene–BAF60C–in cell cultures and in laboratory mice.
Along with the gene HDAC5, BAF60C is thought to indirectly activate the enzyme AKT and promotes glycolysis in muscle cells.
Some of these mice were fed a high-fat diet to induce obesity. Later, they developed trouble disposing of the additional glucose after a meal.
The currently accepted insulin mechanism was not sufficient to process the glucose on its own.
“We found that the molecular pathway that’s engaged by glucose in muscle cells, at least the initial steps, is very similar to what happens in the beta cells in the pancreas,” added Dr Lin, who is also a professor of cell and developmental biology at the University of Michigan Medical School.
Scientifically Proven Method to Reverse Diabetes (Ad)
“It’s amazing how subtle changes in glucose can be detected throughout the body,” Lin said. “Beta cells respond, nerve cells respond, and now we know that muscle cells respond directly, too.”
T2DM in Europe
Metabolic syndrome has become a global epidemic that markedly increases the risk for T2DM, cardiovascular disease, and non-alcoholic steatohepatitis.
T2DM is one of the fastest growing diseases in Europe with about 60 million people currently afflicted.
The World Health Organization (WHO) projects diabetes deaths will double between 2005 and 2030.
Being overweight or obese is an important risk factor for diabetes. It is estimated that about 10.3% of men and 9.6% of women aged 25 years and over in the European Region have the condition.
Impaired muscle insulin action is an early hallmark of obesity-associated metabolic disorders.
 Skeletal muscle insulin resistance has been associated with reduced mitochondrial oxidative function, and a shift from oxidative to glycolytic muscle fibre type in patients with T2DM.
Skeletal muscle insulin resistance has been associated with reduced mitochondrial oxidative function, and a shift from oxidative to glycolytic muscle fibre type in patients with T2DM.
“The engagement of this nutrient-sensing pathway results in glucose-stimulated AKT activation that acts with insulin to promote muscle glucose uptake,” the study concluded.
“As such, muscle-specific transgenic activation of this pathway improved postprandial glucose clearance, whereas its deficiency impaired insulin action, leading to glucose intolerance.”
Natasha Longo writes for PreventDisease.com, where this article first appeared.