Pharmaceutical Drug Trial Industry in Kenya
In January of 2014, the Netherlands-based advocacy group Wemos published a 58-page report “The Clinical Trials Industry in Kenya: Realities, Risks and Challenges” wherein it questions if trial participants are fully informed before they give their consent to participate in new pharmaceutical drug trials, which have become a flourishing industry in Kenya.
African continent countries are becoming “hot” drug trial markets for Phase I, II, and III drug trials, especially Kenya and South Africa. However, there are grave concerns about the safety, effective regulations, and ethical standards of trials conducted by international pharmaceutical companies.
Ambrose Rachier, Head of KEMRI’s ethics committee says, “We look at any compensation that is supposed to be given (to the participants) because when it is very attractive then we are dealing with the principle of inducement – that the people may not be acting in a voluntary manner; they may be coming in for money.”
Widget not in any sidebars
Drug trials are overseen and conducted by two main organizations: the Kenya Medical Research Institute (KEMRI) and U.S. Centers for Disease Control and Prevention (CDC). Major international pharmaceutical companies also are participating more frequently.
Pharmaceutical companies now conducting drug trials in Kenya include: Glaxo Smith Kline (GSK) of the UK; Astra Zeneca, Eli Lilly, and Pfizer of the USA; Sanofi Pasteur of France, Novartis of Switzerland, Bayer and Boehringer Ingelheim of Germany. Kenya is considered, and poised to be, an emerging player in the global trials industry.
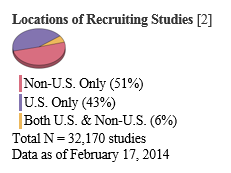
One website that tracks drug trials is ClinicalTrials.gov, which currently lists 161,319 studies taking place in all 50 U.S. states and 185 countries.
That same website also indicates where recruiting studies are going on.
So, pharmaceutical trials really are big business. Another chart indicates the progression of Registered Studies With Posted Results [2] as:
In Kenya currently, there are more than 200 trials taking place throughout the country. Problems occur because participants apparently are not being told the facts of what they are involved with or don’t understand what’s involved. One example is found in what happened to a Kisumu citizen, Edwin.
According to Edwin, “They called me from the line and then they said that they knew I had TB and that I should think about trying a treatment. They said if I wanted to get better without paying anything, I should take the medication, so I just agreed. The other option was to pay five shillings for every hospital visit, and I couldn’t afford it, so it was my only choice.”
…[H]e was asked to sign a consent form in a hospital he visited for TB treatment. The doctor also signed as a witness. “They just said it was a free treatment that would cure me and that some of my blood samples would go to Atlanta in America,” he said.
Edwin says weeks after getting the test drug, he began having strange joints and eye problems. “Slowly my eyesight started to get worse and now, six months later, I really have trouble seeing far away, which is affecting my work,” he said. [1]
It should be noted that problems with eyesight were listed on the consent form that Edwin signed, but probably because of his inability to comprehend what he was signing, he did not understand the full ramifications of a drug trial. According to Edwin, “They will say my eyes are damaged because I go to smoky places.” Edwin was given the drug Rifapentine in doses to expedite a faster cure of TB.
The trial, in which Edwin was a participant, aimed, according to the title on the consent form, to establish efficacy, tolerability, and safety of higher dosage of Rifapentine for TB. [6] [CJF emphasis added]
Apparently, there are many inequities drug trial participants have to deal with, including having to purchase trial drugs at some point in time. “The report says patients who responded well during the trials are cut off from treatment unless they buy the drugs.” [1]
In the Wemos report we find a possible indicator as to why pharmaceutical companies seek out drug trial participants in Kenya:
As the political and public debates continue, the harsh reality of inequality and poverty remain clearly visible in Kenya in 2013. As Erik Okioma, a community health activist and coordinator of the Kenya branch of the AIDS Candlelight Memorial in Kisumu, says frankly: “In Kenya, if you don’t have money and you get seriously ill, you will die.”
It is in this context and against this background of mass poverty and inequality that an increasing number of clinical trials are taking place in Kenya. [3]
Africa has arguably the least access to quality care, ensuring a steady stream of dedicated patients to fill trial enrollments. [4]
 Another example with drug trial problems, as pointed out in the Wemos report, concerns
Another example with drug trial problems, as pointed out in the Wemos report, concerns
Awareness of adverse effects and informed consent
The Declaration of Helsinki states: “In medical research involving human subjects capable of giving informed consent, each potential subject must be adequately informed of the aims, methods, sources of funding, any possible conflicts of interest, institutional affiliations of the researcher, the anticipated benefits and potential risks of the study and the discomfort it may entail, post-study provisions and any other relevant aspects of the study.”24 However, as mentioned before, in the case of Suzanne it seems that she was not informed about the possible side effects of Nevirapine nor was she informed of the risk of HIV transmission as a result of breastfeeding. [5]
Here’s another indication of how drug trials are conducted:
The study, behaviour/observational study on HIV, involved monitoring the sexual activity of highschool age adolescents and taking samples of blood and urine. An investigating team from KEMRI was deployed to the trial site and discovered the consent for the participants had been granted by the head teacher only and not by the parents, a clear ethical violation. [7]
Dr. Patrick Orege of KEMRI’s division in the port city of Kisumu says, “They (sponsors) think they can get away with anything because it’s Africa.” Ambrose Rachier contends, “The public health risk there is obvious.” But here’s probably what may be the cruel and calculated strategy of drug trials, no matter where they are conducted or the pharmaceutical involved:
Investigators don’t always report failure or adverse effects to the sponsor.[8]
Personally and as a researcher, I’d like to have access to the studies and files that pharmaceutical companies – vaccine makers, in particular – do not present to the U.S. FDA during the application for licensure process. I only can imagine what surprises might be found.
Notes:
[1] http://allafrica.com/stories/201402170903.html?viewall=1
[2] http://clinicaltrials.gov/ct2/resources/trends
[3] http://www.wemos.nl/files/Documenten%20Informatief/Bestanden%20voor%20’Medicijnen’/Boekje%20Kenia%20(1).pdf pg. 16
[4] Ibid. pg. 17
[5] Ibid. pg. 27
[6] Ibid. pg. 30
[7] Ibid. pg. 37
[8] Ibid. pg. 37
Catherine J Frompovich (website) is a retired natural nutritionist who earned advanced degrees in Nutrition and Holistic Health Sciences, Certification in Orthomolecular Theory and Practice plus Paralegal Studies. Her work has been published in national and airline magazines since the early 1980s. Catherine authored numerous books on health issues along with co-authoring papers and monographs with physicians, nurses, and holistic healthcare professionals. She has been a consumer healthcare researcher 35 years and counting.
Catherine’s latest book, published October 4, 2013, is Vaccination Voodoo, What YOU Don’t Know About Vaccines, available on Amazon.com.
Her 2012 book A Cancer Answer, Holistic BREAST Cancer Management, A Guide to Effective & Non-Toxic Treatments, is available on Amazon.com and as a Kindle eBook.
Two of Catherine’s more recent books on Amazon.com are Our Chemical Lives And The Hijacking Of Our DNA, A Probe Into What’s Probably Making Us Sick (2009) and Lord, How Can I Make It Through Grieving My Loss, An Inspirational Guide Through the Grieving Process (2008).